 
The molecules in liquids are closer together than the molecules in gases. When you pour water from a pitcher into a glass, the water flows from the pitcher into the glass. This means that they can be poured from one container to another. This means that if you pour water into a cup, it will take the shape of the cup. A liquid has a definite shape, but it takes the shape of its container. For example, a solid has a definite shape and a gas does not have a definite shape. Liquids have different properties than solids and gases. All liquids are made up of atoms, which are held together by forces called van der Waals forces. What Are Liquids?Ī liquid is a material that has a definite shape but takes the shape of its container. The forces between the atoms can be either ionic, covalent, or metallic bonding. Atoms in solids are closely packed together and are held in place by strong forces between the atoms. Unlike liquids, solids cannot be easily deformed. Solids are matter that has a definite shape and volume. If you put the glass of cold water in the refrigerator, the water will eventually freeze and become ice again. For example, when you put ice in a glass of water, the ice will melt and the water will become cold. The states of matter are always changing. When water changes from a liquid to a gas, we say that it boils. When water changes from a solid to a liquid, we say that it melts. For example, water can exist as a solid (ice), a liquid (water), or a gas (steam). When matter changes state, it means that it changes from one form to another. This is what gives gases their low densities and makes them easy to compress. In gases, the particles are far apart with very weak attractive forces between them. The attractive forces between them are weaker than in solids, so liquids can flow around each other easily. In liquids, the particles are closer together than in gases, but not as close as in solids. This gives solids their rigidity, or resistance to being deformed. In a solid, the particles are close together with strong attractive forces between them. The difference is in how much space the particles have between them, and how tightly they are held together by attractive forces. The particles in all three states are always moving around. They do not have a definite shape or volume. Gases are the most energetic state of matter. They flow and take the shape of their container. They have a definite volume, but they do not have a definite shape. Solids are the least energetic state of matter. 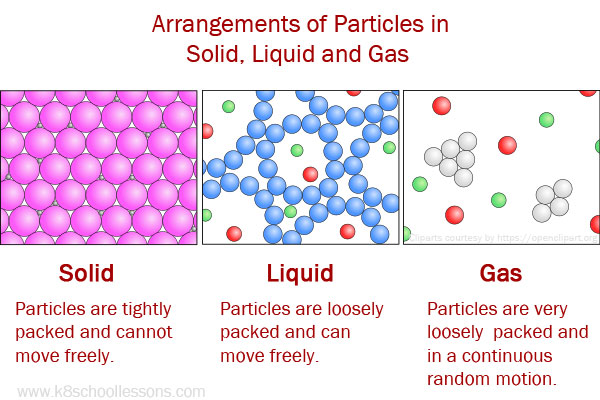
There are three states of matter: solid, liquid, and gas. Gases are the most reactive state of matter and have no definite shape or volume. Liquids are more reactive than solids and have a definite volume but no definite shape. Solids are the least reactive state of matter and are characterized by a definite shape and volume. Solids, liquids, and gases are all states of matter. Adding more energy can change a gas into a plasma, which is a fourth state of matter. When cooled, it changes back to a liquid. For example, when water is heated, it changes from a liquid to a gas (steam). 
The states of matter can be changed by adding or removing energy. Gases are the most compressible state of matter and do not have a fixed shape or volume. Liquids are more compressible than solids and have a fixed volume, but they take the shape of their container. This means that they have a fixed shape and volume. Solids are the least compressible state of matter. Each state has its own unique properties. The three states of matter are: solid, liquid, and gas. In order to understand solids, liquids, and gases, it is important to first understand the states of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
